Introduction: Currently, there are many 2nd-line treatment regimens for relapsed Multiple Myeloma (MM) but no standard therapy. Daratumumab, pomalidomide, and dexamethasone (DPd) is a newer 3-drug regimen approved by the FDA for treatment of multiple myeloma in the 3rd or later lines.
The POLLUX trial reported a 12-month PFS of 83% in relapsed (median of one prior treatment line) MM treated with daratumumab, lenalidomide, and dexamethasone but excluded lenalidomide-refractory patients. A meta-analysis by Premkumar et al recently showed that high risk MM, del17p, t(4:14), t(14:16) cytogenetics, had minimal benefit from daratumumab-based therapies as 1st-line but benefited more in the 3rd-line or later setting. Nooka et al. previously reported increased response in daratumumab and pomalidomide naïve patients with relapsed refractory MM: median PFS of 41 months in a cohort of 12 patients.
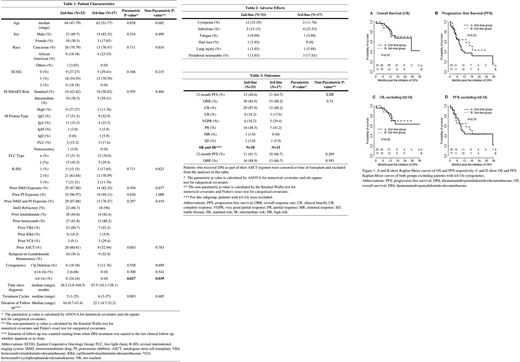
A recent Phase II trial by Siegel et al. demonstrated decreased efficacy of 2nd and 3rd-line DPd (1-year PFS of 45.2% vs 82.8% and ORR of 55.0% and 79.5%) in high risk versus standard risk patients respectively. However, it is unclear whether this pattern is consistent between patients treated with DPd in the 2nd vs 3rd-line. Herein, we report the efficacy of DPd when used in the 2nd versus 3rd-line depending on patient MSMART risk category.
Methods: We reviewed pharmacy and institutional records of patients who began treatment with DPd in the 2nd (n = 33) or 3rd-line (n = 17) from April 2016 to March 2019. Patients had at least 1 year of follow up from starting DPd unless they progressed or expired before then. A line of therapy was defined as the therapy received between the events of diagnosis, progression, and/or death. We compared the ORR and 12-month PFS of 2nd-line and 3rd-line DPd. The differences in the 12-month PFS and ORR were compared using Fisher's exact test. Odds ratios (OR) were calculated from univariate/multivariate logistic regressions.
Results: Thirty-three patients (23 men and 10 women), with median age of 63 (range 47 - 79) and median ECOG of 1, were treated with DPd as 2nd-line therapy. The 3rd-line DPd group was similar, consisting of 17 patients (14 men and 3 women), with median age of 62 (range 51 - 77) and median ECOG of 1. One patient was excluded from analysis in the 2nd-line group due to loss to follow up. Six patients were censored at time of transplant in the 2nd-line group: 4 (12.1%) received DPd as induction therapy for ASCT and 2 (6.1%) received DPd as maintenance therapy after ASCT. In the 3rd-line group, 2 (11.8%) received DPd as induction therapy and were censored. The most common side effects were cytopenias (35.3%), infections (15.2%), fatigue (8.8%). Most of the patients were daratumumab and pomalidomide naïve except one patient in the 3rd-line DPd group who had prior pomalidomide exposure. Twenty-two (66.7%) patients in the 2nd-line group were IMiD refractory versus 16 (94%) in the 3rd-line group. The 12-month PFS for the 2nd-line group was 40.6% compared with 64.7% in the 3rd-line group and showed a trend towards statistical significance (OR=2.82, p=0.09), and the difference reduced (OR=1.49, p=0.57) after adjusting for M-SMART risk category and t(4:14) cytogenetics. On exclusion of high risk and t(4:14), the 12-month PFS was 61.1% vs 66.7% for 2nd- and 3rd-line respectively (p>0.99). The ORR was 84.9% in the 2nd-line group and 82.2% in the 3rd-line group (OR=1.34, p=0.74). The median follow-up for survivors were 22.3 months (range 2.5-43.4). 30 patients relapsed and 16 patients died during follow-up period. M-SMART high-risk designation (HR 2.56; 95%CI 1.09-6.04) and t(4:14) cytogenetics (HR 3.12; 95%CI 1.32-7.43) were associated with lower PFS. Older age of diagnosis was associated with a lower OS (HR 1.11; 95%CI 1.03-1.20).
Conclusion: The difference in length of PFS between 2nd and 3rd- line DPd is likely an artifact of small sample size and differential efficacy of DPd depending on cytogenetics. Our results show comparable efficacy of 2nd to 3rd-line DPd when used in standard risk and non-t(4:14) intermediate risk MM. In patients with high risk or t(4:14) cytogenetics, it may be preferable to use DPd in the 3rd or later line.
Goldsmith:Wugen Inc.: Consultancy. Wildes:Carevive Systems: Consultancy; Janssen: Research Funding; Seattle Genetics: Consultancy. Schroeder:PBD Incorporated: Research Funding; Janssen: Research Funding; Dova Pharmaceuticals: Other; Astellas: Other; Gilead Sciences Inc: Other; GSK: Other; Celgene: Research Funding; Amgen: Other: served on advisory boards and received honoraria or consultant fees, Research Funding; Takeda: Consultancy, Honoraria, Speakers Bureau; Merck: Consultancy, Honoraria, Speakers Bureau; AbbVie: Consultancy, Honoraria, Speakers Bureau; Pfizer: Other; Genzyme Sanofi: Other: served on advisory boards and received honoraria or consultant fees, Research Funding; Partners Therapeutics: Other; Novo Nordisk: Other; Seattle Genetics: Research Funding; Fortis: Research Funding; Cellect Inc: Research Funding; Incyte Corporation: Other: served on advisory boards and received honoraria or consultant fees, Research Funding; Genentech Inc: Research Funding; FlatIron Inc: Other.
Daratumumab, pomalidomide, and dexamethasone is approved for treatment of relapsed, refractory multiple myeloma.
Author notes
Asterisk with author names denotes non-ASH members.